Research
Crystal Design & MOF Synthesis | Detoxification of Chemical Warfare Agents | Enzyme Encapsulation
Actinide-based MOFs | Heterogeneous Catalysis for Energy-Related Applications | Water Purification | Gas Storage/Separation
Chemical Separation | Physical and Chemical Stability of MOFs | Characterization Tools for MOFs | Porous Organic Polymers
Porous Clusters | Composite Materials
Crystal Design & MOF Synthesis
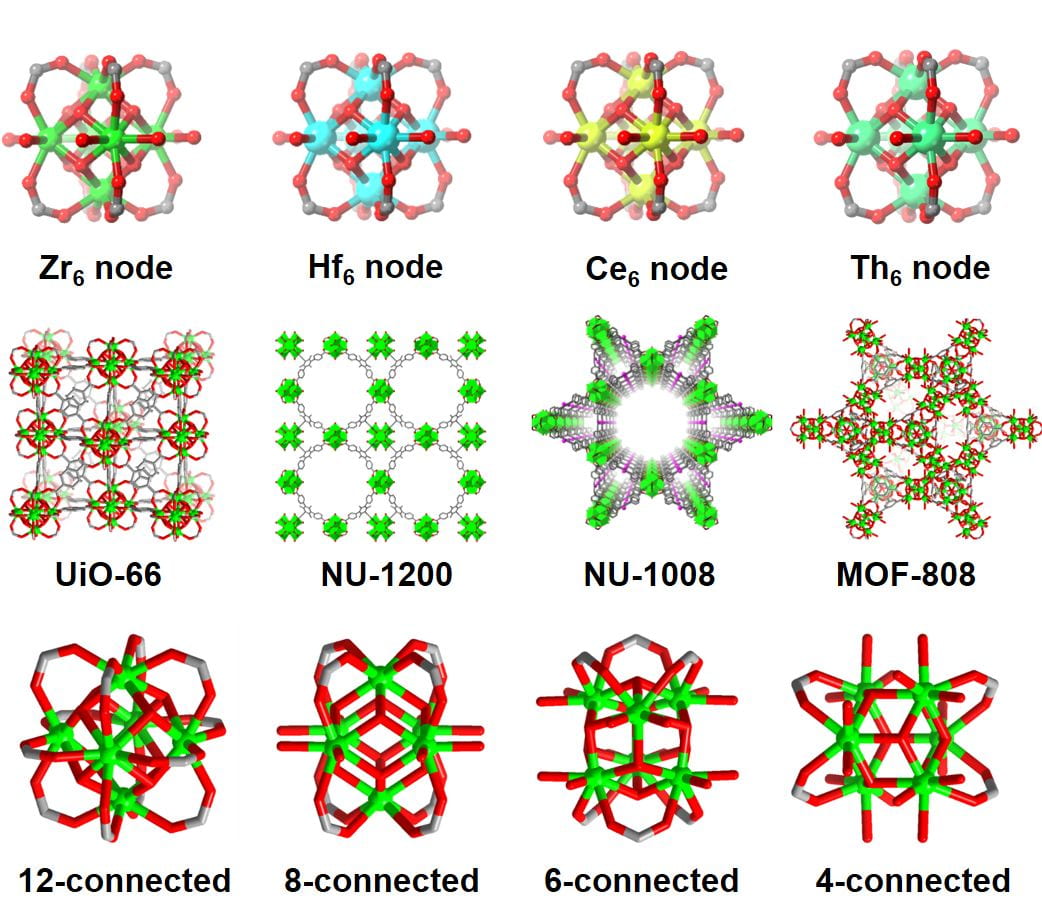
While there are many serendipitous discoveries in MOF chemistry, rational top-down design and assembly with molecular-level precision is crucial for the efficient preparation of novel, targeted MOF materials. The expansion of topology-directed crystal engineering, referred to as reticular chemistry, permits the geometry-guided architectural arrangement of crystalline frameworks (e.g. MOFs) by linking pre-selected molecular building blocks (MBBs). The applications of topology-directed crystal engineering can be divided into two parts: the discovery of novel materials and the execution of isoreticular chemistry. The former generally aims to design a blueprint net as a target, and then assemble pre-selected organic and inorganic MBBs to achieve the desired topology. The latter, on the other hand, aims to fine tune and functionalize the MBBs of a preexisting MOF to achieve structural analogues with unique and desirable properties. While their modular nature allows for designing functional MOFs using de novo synthesis, there are conditions where desired functionality is very challenging to introduce using direct synthetic methods. In these cases, we have developed post-synthesis crystal engineering methods to access preferred functionalities. These techniques include methods for functionalizing MOF nodes, i.e., solvent-assisted ligand incorporation (SALI) and atomic layer deposition in MOFs (AIM) as well as solvent-assisted linker exchange (SALE), a method to replace structural linkers. We continue to pioneer novel synthetic methodologies which allow us to access atomically precise MOFs with novel topological properties.
Selected Reviews
- “The state of the field: from inception to commercialization of metal–organic frameworks.” Chen, Z.; Wasson, M. C.; Drout, R. J.; Robison, L.; Idrees, K. B.; Knapp, J. G.; Son, F. A.; Zhang, X.; Hierse, W.; Kühn, C.; Marx, S.; Hernandez, B.; Farha, O. K. Faraday Discuss. 2020. doi.org/10.1039/D0FD00103A
- “A historical overview of the activation and porosity of metal–organic frameworks.” Zhang, X.; Chen, Z.; Liu, X.; Hanna, S. L.; Wang, X.; Taheri-Ledari, R.; Maleki, A.; Li, P.; Farha, O. K. Chem. Soc. Rev. 2020, 49, 7406-7427. doi.org/10.1039/D0CS00997K
- ” A historical perspective on porphyrin-based metal–organic frameworks and their applications.” Zhang, X.; Wasson, M. C.; Shayan, M.; Berdichevsky, E. K.; Ricardo-Noordberg, J.; Singh, Z.; Papazyan, E. K.; Castro, A. J.; Marino, P.; Ajoyan, Z.; Chen, Z.; Islamoglu, T.; Howarth, A. J.; Liu, Y.; Majewski, M. B.; Katz, M. J.; Mondloch, J. E.; Farha, O. K. Coord. Chem. Rev. 2020, 213615. doi.org/10.1016/j.ccr.2020.213615
- “Reticular Chemistry in the Rational Synthesis of Functional Zirconium Cluster-Based MOFs” Chen, Z.; Hanna, S. L.; Redfern, L. R.; Alezi, D.; Islamoglu, T.; Farha, O. K. Coord. Chem. Rev. 2019, 386, 32-49. doi.org/10.1016/j.ccr.2019.01.017
Selected Research Articles
-
“Topological Strain-Induced RegioselectiveLinker Elimination in a Chiral Zr(IV)-BasedMetal-Organic Framework” Gong, W.; Zhang, W.; Son, F.A.; Yang, K.; Chen, Z.; Chen, X.; Jiang, J.; Liu, Y.; Farha, O.K.; Cui, Y. Chem, 2021, 7, pp. 190-201 doi.org/10.1016/j.chempr.2020.11.008
-
“Highly Specific Coordination-Driven Self-Assembly of 2D Heterometallic Metal–Organic Frameworks with Unprecedented Johnson-type (J51) Nonanuclear Zr-Oxocarboxylate Clusters” Gong, W.; Arman, H.; Chen, Z.; Xie, Y.; Son, F.A.; Cui, H.; Chen, X.; Shi, Y.; Liu, Y.; Chen, B.; Farha, O.K.; Cui, Y. J. Am. Chem. Soc., 2021, 143, pp. 657-663 doi.org/10.1021/jacs.0c11881
-
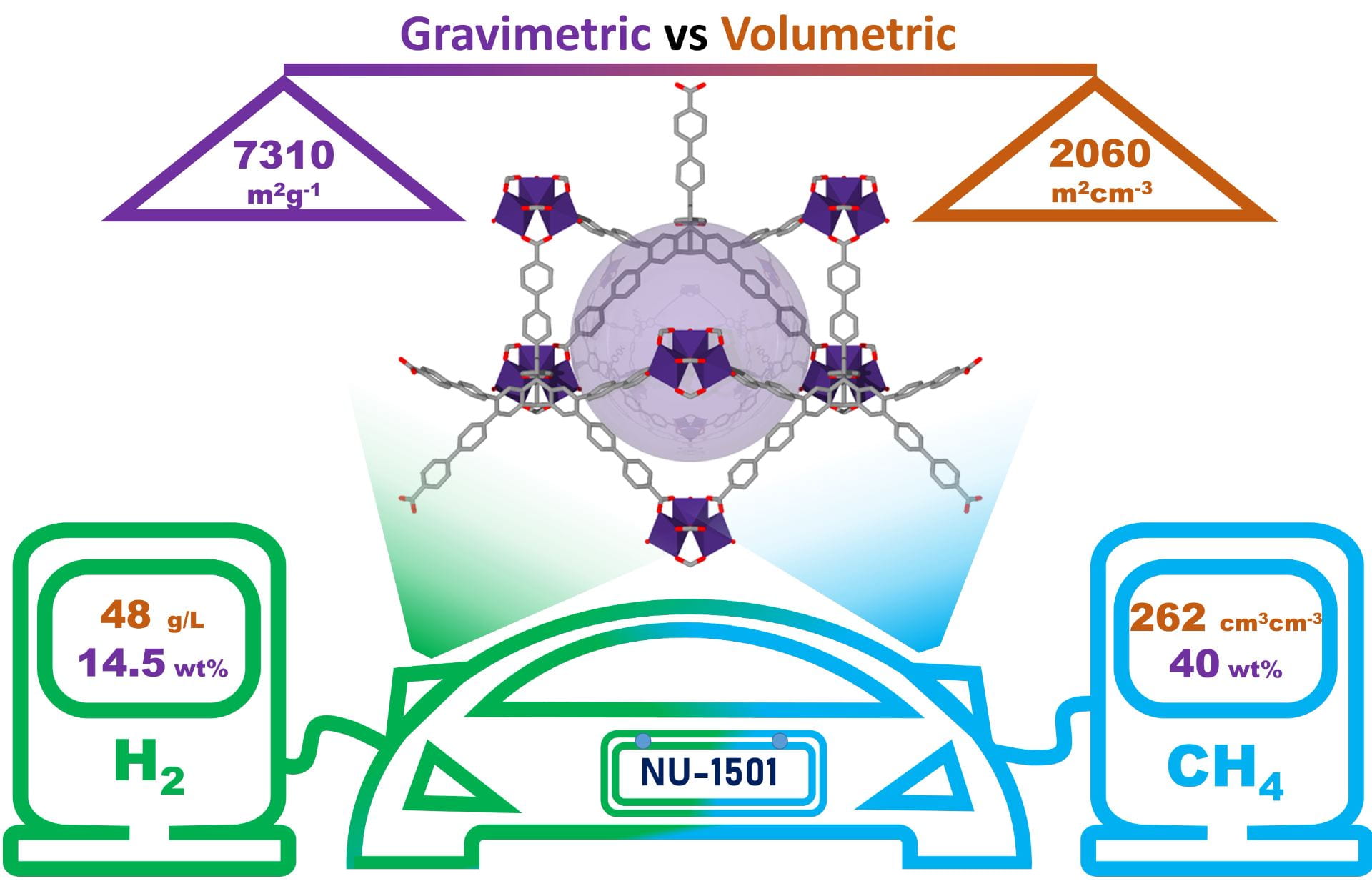
“Balancing volumetric and gravimetric uptake in highly porous materials for clean energy” Chen, Z.; Li, P.; Anderson, R.; Wang, X.; Zhang, X.; Robison, L.; Redfern, L.R.; Moribe, S.; Islamoglu, T.; Gómez-Gualdrón, D.A.; Yildirim, T.; Stoddart, J.F.; Farha, O.K.; Science, 2020, 368, pp. 297-303 doi.org/10.1126/science.aaz8881
-
“Structural Diversity of Zirconium Metal–Organic Frameworks and Effect on Adsorption of Toxic Chemicals” Chen, Y.; Xuan, Z.; Mian, M.R.; Son, F.A.; Zhang, K.; Cao, R.; Chen, Z.; Lee, S.-J.; Idrees, K.B.; Goetjen, T.A.; Lyu, J.; Li, P.; Xia, Q.; Li, Z.; Hupp, J.T.; Islamoglu, T.; Napolitano, A.; Peterson, G.W.; Farha, O.K. J. Am. Chem. Soc., 2020, 142, pp. 21428-21438 doi.org/10.1021/jacs.0c10400
- “Ligand-Directed Reticular Synthesis of Catalytically Active Missing Zirconium-Based Metal–Organic Frameworks” Chen, Z.; Li, P.; Wang, X.; Otake, K.-i.; Zhang, X.; Robison, L.; Atilgan, A.; Islamoglu, T.; Hall, M. G.; Peterson, G. W.; Stoddart, J. F.; Farha, O. K. Am. Chem. Soc. 2019, 141, 12229-12235. doi.org/10.1021/jacs.9b06179
- “Topology and Porosity Control of Metal-Organic Frameworks through Linker Functionalization” Lyu, J. F.; Zhang, X.; Otake, K.; Wang, X. J.; Li, P.; Li, Z. Y.; Chen, Z. J.; Zhang, Y. Y.; Wasson, M. C.; Yang, Y.; Bai, P.; Guo, X. H.; Islamoglu, T.; Farha, O. K. Sci. 2019, 10, 1186-1192. DOI:10.1039/C8SC04220A
- “Bottom-up construction of a superstructure in a porous uranium-organic crystal” Li, P.; Vermeulen, N. A.; Malliakas, C. D.; Gómez-Gualdrón, D. A.; Howarth, A. J.; Mehdi, B. L.; Dohnalkova, A.; Browning, N. D.; O’Keeffe, M.; Farha, O. K., Science 2017,356, 624-627. org/10.1126/science.aam7851
Detoxification of Chemical Warfare Agents
Organophosphonate-based Nerve Agents
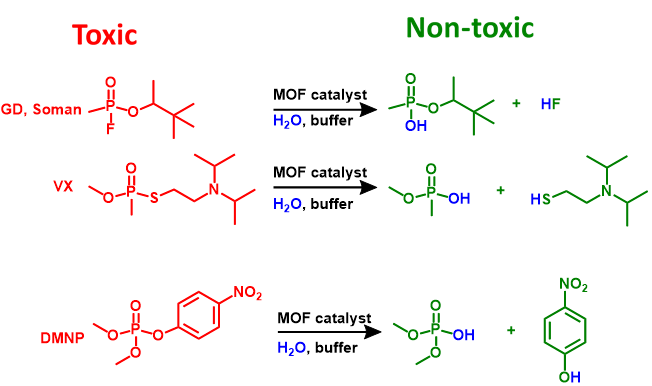 Chemical warfare agents containing phosphonate ester bonds are among the most toxic chemicals ever created. Solutions are needed for immediate personal protection (for example, the filtration and catalytic destruction of airborne versions of agents), bulk destruction of chemical weapon stockpiles, protection (via coating) of clothing, equipment and buildings, and containment of agent spills. The selective, hydrolytic cleavage of the P—F bond of organophosphonate based nerve agents is effective for chemical detoxification of these agents. P—F bond cleavage with cationic Lewis acids is particularly promising as phosphonate ester hydrolysis can be accelerated by polarizing the P═O bond.
Chemical warfare agents containing phosphonate ester bonds are among the most toxic chemicals ever created. Solutions are needed for immediate personal protection (for example, the filtration and catalytic destruction of airborne versions of agents), bulk destruction of chemical weapon stockpiles, protection (via coating) of clothing, equipment and buildings, and containment of agent spills. The selective, hydrolytic cleavage of the P—F bond of organophosphonate based nerve agents is effective for chemical detoxification of these agents. P—F bond cleavage with cationic Lewis acids is particularly promising as phosphonate ester hydrolysis can be accelerated by polarizing the P═O bond.
In the Farha group, we design/identify metal-organic frameworks (MOFs) that can effectively catalyze the important reactions shown above. We fine tune chemical (i.e. metal, functional groups etc.) and/or physical properties (node connectivity, topology, pore size, particle size etc.) of the MOFs and monitor the reactivity towards a nerve agent simulant, DMNP, in our lab, and towards actual nerve agents such as VX and GD in collaboration with US. Army Research labs. We have developed materials that show almost instantaneous hydrolysis of these toxic chemicals using catalytic amounts of MOFs.
Selected Reviews
- “Zirconium-Based Metal-Organic Frameworks for the Catalytic Hydrolysis of Organophosphorus Nerve Agents” Kirlikovali, K.O.; Chen, Z.; Islamoglu, T.; Hupp, J.T.; Farha, O.K.; ACS Appl. Mater. Interfaces, 2020, 12, pp. 14702-14720 doi.org/10.1021/acsami.9b20154
- “Metal-Organic Frameworks against Toxic Chemicals” Islamoglu, T.; Chen, Z.; Wasson, M.C.; Buru, C.T.; Kirlikovali, K.O.; Afrin, U.; Mian, M.R.; Farha, O.K.; Chem. Rev., 2020, 120, pp. 8130-8160 doi.org/10.1021/acs.chemrev.9b00828
- “Metal-organic frameworks for the removal of toxic industrial chemicals and chemical warfare agents.” Bobbitt, N. S.; Mendonca, M. L.; Howarth, A. J.; Islamoglu, T.; Hupp, J. T.; Farha, O. K.; Snurr, R. Q.; Soc. Rev.; 2017, 46, pp.3357-3385. doi.org/10.1039/C7CS00108H
-
“Catalytic degradation of chemical warfare agents and their simulants by metal-organic frameworks” Liu, Y.; Howarth, A. J.; Vermeulen, N. A.; Moon, S.-Y.; Hupp, J. T.; Farha, O. K.; Coord. Chem. Rev., 2017, 346, pp. 101-111 doi.org/10.1016/j.ccr.2016.11.008
Selected Research Articles
-
“Near-instantaneous catalytic hydrolysis of organophosphorus nerve agents with zirconium-based metal-organic-framework hydrogel composites” Ma, K.; Wasson, M.C.; Wang X.; Zhang X.; Idrees, K.B.; Chen, Z.; Wu, Y.; Lee, S.-J.; Cao, R.; Chen, Y.; Yang, L.; Son, F.A.; Islamoglu, T.; Peterson, G.W.; Mahle, J.J.; Farha, O.K. Chem Catalysis, 2021, 1, pp. 1-13 doi.org/10.1016/j.checat.2021.06.008
-
“Insights into Catalytic Hydrolysis of Organophosphonates at M–OH Sites of Azolate-Based Metal Organic Frameworks” Mian, M. R.; Chen, H.; Cao, R.; Kirlikovali, K. O.; Snurr, R. Q.; Islamoblu, T.; Farha, O.K. J. Am. Chem. Soc., 2021, 143, pp. 9893-9900 doi.org/10.1021/jacs.1c03901
- “Integration of Metal–Organic Frameworks on Protective Layers for Destruction of Nerve Agents under Relevant Conditions” Chen, Z.; Ma, K.; Mahle, J. J.; Wang, H.; Syed, Z. H.; Atilgan, A.; Chen, Y.; Xin, J. H.; Islamoglu, T.; Peterson, G. W.; Farha, O. K., Am. Chem. Soc. 2019, 141, (51), 20016-20021. doi.org/10.1021/jacs.9b11172
- “Scalable and Template-Free Aqueous Synthesis of Zirconium-Based Metal–Organic Framework Coating on Textile Fiber.” Ma, K.; Islamoglu, T.; Chen, Z.; Li, P.; Wasson, M.C.; Chen, Y.; Wang, Y.; Peterson, G.W.; Xin, J.H.; Farha, O.K.; Am. Chem. Soc.; 2019, 141, pp. 15626-15633. doi.org/10.1021/jacs.9b07301
- “Presence versus Proximity: The Role of Pendant Amines in the Catalytic Hydrolysis of a Nerve Agent Simulant.” Islamoglu, T.; Ortuño, M. A.; Proussaloglou, E.; Howarth, A. J.; Vermeulen, N. A.; Atilgan, A.; Asiri, A. M.; Cramer, C. J.; Farha, O. K.; Chem. Int. Ed.; 2018, 130, pp. 1967-1971.
doi.org/10.1002/anie.201712645 - “Cerium(IV) vs Zirconium(IV) Based Metal-Organic Frameworks for Detoxification of a Nerve Agent.” Islamoglu, T.; Atilgan, A.; Moon, S. Y.; Peterson, G. W.; DeCoste, J. B.; Hall, M.; Hupp, J. T.; Farha, O. K., Chem. Mater. 2017, 29, (7), 2672-2675. doi.org/10.1021/acs.chemmater.6b04835
Detoxification of HD and its simulants
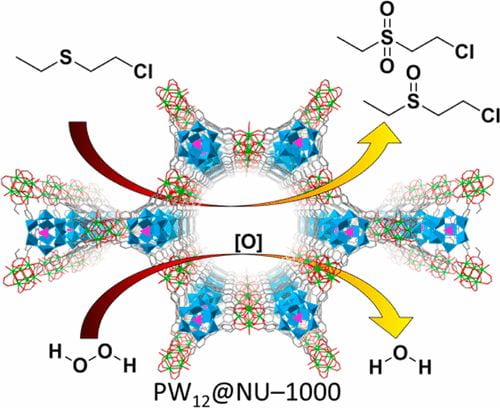
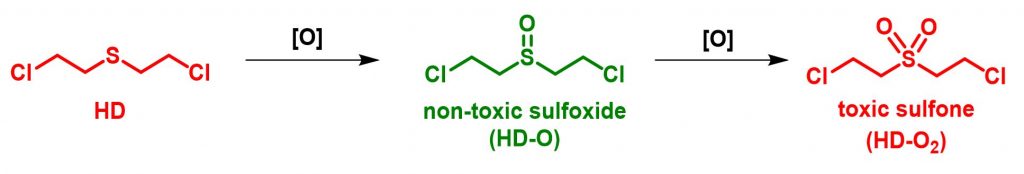 The continued prevalence of chemical warfare agents (CWAs) as weapons of mass destruction (WMD) and in stockpile materials necessitates the development of materials capable of rapid detoxification of these chemicals.Among the commonly used CWAs, sulfur mustard (also known as mustard gas or HD) incapacitates its victims by causing painful blisters and irreversible tissue damage. We have found oxidation of HD to the nontoxic sulfoxide derivative is a viable detoxification pathway. The challenge of this oxidation occurs with over-oxidation to the sulfone derivative, which has similar toxic properties as the parent HD; therefore, the reaction must selectively yield the sulfoxide.
The continued prevalence of chemical warfare agents (CWAs) as weapons of mass destruction (WMD) and in stockpile materials necessitates the development of materials capable of rapid detoxification of these chemicals.Among the commonly used CWAs, sulfur mustard (also known as mustard gas or HD) incapacitates its victims by causing painful blisters and irreversible tissue damage. We have found oxidation of HD to the nontoxic sulfoxide derivative is a viable detoxification pathway. The challenge of this oxidation occurs with over-oxidation to the sulfone derivative, which has similar toxic properties as the parent HD; therefore, the reaction must selectively yield the sulfoxide.
In the Farha group, we have taken two approaches for the rapid, selective oxidation of HD: 1) using MOF linkers and ligands as photosensitizers to generate singlet oxygen from atmospheric oxygen upon LED irradiation and 2) installing soluble, but catalytically active polyoxometalates (POMs) in a MOF to activate hydrogen peroxide or atmospheric oxygen. At Northwestern, we test these materials using simulants, chemicals with similar properties as CWA but much less toxic; our collaborators have capabilities allowing them to test our materials against the real agents.
Because the linkers and the installation of POMs leave the MOF node untouched, HD oxidation can be coupled with the decomposition of toxic organophosphate nerve agents, a reaction which occurs on the MOF nodes. We wish to continue developing new materials and composites to achieve the ultimate catalyst for CWA detoxification.
Selected Research Articles
- “Restricting Polyoxometalate Movement within Metal-Organic Frameworks to Assess the Role of Residual Water in Catalytic Thioether Oxidation Using These Dynamic Composites” Buru, C. T.; Lyu, J.; Liu, J.; Farha, O. K. Frontiers in Materials 2019, 6, 152. doi.org/10.3389/fmats.2019.00152
- “Thermally Induced Migration of a Polyoxometalate within a Metal-Organic Framework and Its Catalytic Effects” Buru, C. T.; Platero-Prats, A. E.; Chica, D. G.; Kanatzidis, M. G.; Chapman, K. W.; Farha, O. K. J. Mater. Chem. A 2018, 6, 7389-7394. doi:10.1039/c8ta02562b
- “Postsynthetic Incorporation of a Singlet Oxygen Photosensitizer in a Metal–Organic Framework for Fast and Selective Oxidative Detoxification of Sulfur Mustard” Howarth, A. J.; Buru, C. T.; Liu, Y.; Ploskonka, A. M.; Hartlieb, K. J.; McEntee, M.; Mahle, J. J.; Buchanan, J. H.; Durke, E. M.; Al-Juaid, S. S.; Stoddart, J. F.; DeCoste, J. B.; Hupp, J. T.; Farha, O. K., Chem. Eur. J. 2017, 23, 214-218. doi.org/10.1002/chem.201604972
Detoxification of HD and its simulants
 The continued prevalence of chemical warfare agents (CWAs) as weapons of mass destruction (WMD) and in stockpile materials necessitates the development of materials capable of rapid detoxification of these chemicals.Among the commonly used CWAs, sulfur mustard (also known as mustard gas or HD) incapacitates its victims by causing painful blisters and irreversible tissue damage. We have found oxidation of HD to the nontoxic sulfoxide derivative is a viable detoxification pathway. The challenge of this oxidation occurs with over-oxidation to the sulfone derivative, which has similar toxic properties as the parent HD; therefore, the reaction must selectively yield the sulfoxide.
The continued prevalence of chemical warfare agents (CWAs) as weapons of mass destruction (WMD) and in stockpile materials necessitates the development of materials capable of rapid detoxification of these chemicals.Among the commonly used CWAs, sulfur mustard (also known as mustard gas or HD) incapacitates its victims by causing painful blisters and irreversible tissue damage. We have found oxidation of HD to the nontoxic sulfoxide derivative is a viable detoxification pathway. The challenge of this oxidation occurs with over-oxidation to the sulfone derivative, which has similar toxic properties as the parent HD; therefore, the reaction must selectively yield the sulfoxide.
In the Farha group, we have taken two approaches for the rapid, selective oxidation of HD: 1) using MOF linkers and ligands as photosensitizers to generate singlet oxygen from atmospheric oxygen upon LED irradiation and 2) installing soluble, but catalytically active polyoxometalates (POMs) in a MOF to activate hydrogen peroxide or atmospheric oxygen. At Northwestern, we test these materials using simulants, chemicals with similar properties as CWA but much less toxic; our collaborators have capabilities allowing them to test our materials against the real agents.
Because the linkers and the installation of POMs leave the MOF node untouched, HD oxidation can be coupled with the decomposition of toxic organophosphate nerve agents, a reaction which occurs on the MOF nodes. We wish to continue developing new materials and composites to achieve the ultimate catalyst for CWA detoxification.
Selected Research Articles
- “Restricting Polyoxometalate Movement within Metal-Organic Frameworks to Assess the Role of Residual Water in Catalytic Thioether Oxidation Using These Dynamic Composites” Buru, C. T.; Lyu, J.; Liu, J.; Farha, O. K. Frontiers in Materials 2019, 6, 152. doi.org/10.3389/fmats.2019.00152
- “Thermally Induced Migration of a Polyoxometalate within a Metal-Organic Framework and Its Catalytic Effects” Buru, C. T.; Platero-Prats, A. E.; Chica, D. G.; Kanatzidis, M. G.; Chapman, K. W.; Farha, O. K. J. Mater. Chem. A 2018, 6, 7389-7394. doi:10.1039/c8ta02562b
- “Postsynthetic Incorporation of a Singlet Oxygen Photosensitizer in a Metal–Organic Framework for Fast and Selective Oxidative Detoxification of Sulfur Mustard” Howarth, A. J.; Buru, C. T.; Liu, Y.; Ploskonka, A. M.; Hartlieb, K. J.; McEntee, M.; Mahle, J. J.; Buchanan, J. H.; Durke, E. M.; Al-Juaid, S. S.; Stoddart, J. F.; DeCoste, J. B.; Hupp, J. T.; Farha, O. K., Chem. Eur. J. 2017, 23, 214-218. doi.org/10.1002/chem.201604972
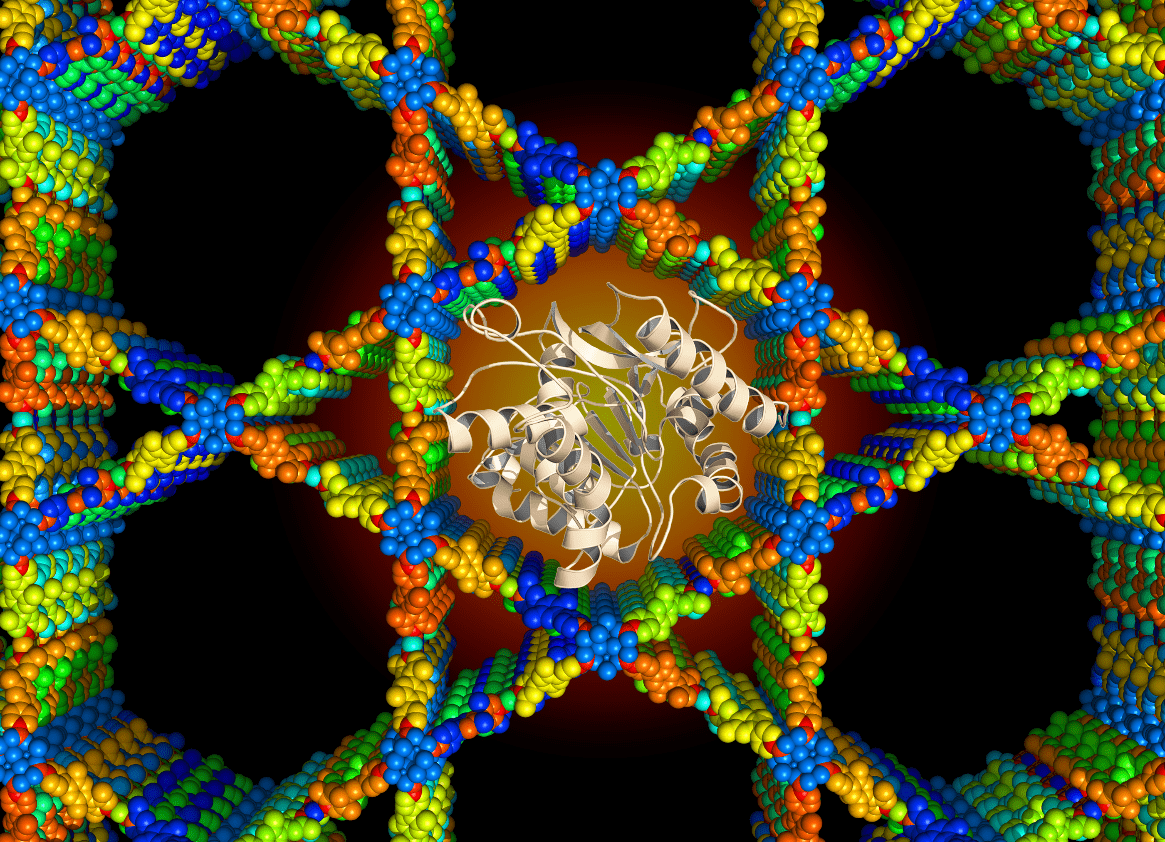
Enzyme Encapsulation
One significant obstacle to using recombinant proteins for biocatalytic and biotechnological applications is the production costs of enzymes and coenzymes in addition to high separation and isolation costs, intolerance to toxic products, and poor thermal and long-term stability. To address these challenges, efficient immobilization strategies based on biocompatible carriers are needed to realize the use of enzymes and regeneration of coenzymes for large-scale industrial production of fine chemicals, drug delivery and biocatalytic applications. The effective use of hybrid materials requires a delicate balance among structure, biocompatibility, and stability. The specific aims in Farha group under this research topic include:
1) Develop efficient de novo and post-modification immobilization strategies to prepare stable and biocompatible inorganic/organic/biological hybrid materials.
2) Study the diffusion, accessibility, and reactivity of biomolecules confined in the nanospace of well-defined porous crystalline materials.
3) Investigate the stability of the encapsulated enzymes and their ability to withstand pH changes, temperature swings and toxic intermediates formed during biocatalysis.
Selected Reviews
- “Catalytic applications of enzymes encapsulated in metal-organic frameworks.” Drout, R.J.; Robison, L.; Farha, O.K.; Coord. Chem. Rev.;2019, 381, pp. 151-160.
doi.org/10.1016/j.ccr.2018.11.009
- “Enzyme Encapsulation in Metal–organic Frameworks for Applications in Catalysis.” Majewski, M. B.; Howarth, A. J.; Li, P.; Wasielewski, M. R.; Hupp, J. T.; Farha, O. K., CrystEngComm 2017, 19, 4082-4091. doi.org/10.1039/C7CE00022G
Selected Research Articles
-
“Stabilization of an enzyme cytochrome c in a metal-organic framework against denaturing organic solvents” Sha, F.; Chen, Y.; Drout, R.J.; Idrees, K.B.; Zhang, X.; Farha, O.K. iScience, 2021, 24, pp. 102641 doi.org/10.1016/j.isci.2021.102641
- “Hierarchically Engineered Mesoporous Metal-Organic Frameworks toward Cell-free Immobilized Enzyme Systems.” Li, P.; Chen, Q.; Wang, T. C.; Vermeulen, N. A.; Mehdi, B. L.; Dohnalkova, A.; Browning, N. D.; Shen, D.; Anderson, R.; Gómez-Gualdrón, D. A.; Cetin, F. M.; Jagiello, J.; Asiri, A. M.; Stoddart, J. F.; Farha, O. K.; Chem; 2018, 4, pp. 1022–1034. doi.org/10.1016/j.chempr.2018.03.001
- “DNA-Functionalized Metal-Organic Framework Nanoparticles for Intracellular Delivery of Proteins.” Wang, S.; Chen, Y.; Wang, S.; Li, P.; Mirkin, C.A.; Farha, O.K.; Am. Chem. Soc.; 2019, 141, pp. 2215-2219. doi.org/10.1021/jacs.8b12705
- “Acid-Resistant Mesoporous Metal–Organic Framework toward Oral Insulin Delivery: Protein Encapsulation, Protection, and Release.” Chen, Y.; Li, P.; Modica, J. A.; Drout, R. J.; Farha, O. K.; Am. Chem. Soc.; 2018, 140, pp. 5678-5681. doi.org/10.1021/jacs.8b02089
Actinide-based MOFs
The subject of actinide-based functional materials is substantially underexplored but is highly intriguing given the unique electronic structure of these elements, which includes 5f electrons. Controlling the bond formation of actinides could lead to materials with unconventional chemical, electronic, and magnetic properties. For example, uranyl compounds exhibit ligand to metal charge transfer (LMCT) in the visible region and are luminescent. By capitalizing on and tailoring these properties, uranium compounds could be utilized for catalytic degradation of water, photodegradation of organic dyes, oxidative combustion of volatile organic compounds, and activation of small molecules such as N2, CO, and CO2. Additionally, the uranium-based heavy fermion compounds exhibit a new type of superconductivity distinct from that of traditional superconductors. Trivalent and pentavalent uranium molecular compounds can display single-molecule magnet behavior with noticeably high relaxation barriers. The specific aims in Farha Group under this research topic include:
1) Synthesize and characterize novel actinides-based materials.
2) Understand the chemical bonding and processes that govern the formation of actinide-based materials.
3) Customize and tailor functional actinides-based materials on demand.
Selected Research Articles
-
“Stabilization of Photocatalytically Active Uranyl Species in a Uranyl–Organic Framework for Heterogeneous Alkane Fluorination Driven by Visible Light” Zhang, X.; Li, P.; Krzyaniak, M.; Knapp, J.; Wasielewski, M.R.; Farha, O.K.; Inorg. Chem., 2020, 59, pp. 16795-16798 doi.org/10.1021/acs.inorgchem.0c00850
- “In Situ Formation of Unprecedented Neptunium-Oxide Wheel Clusters Stabilized in a Metal-Organic Framework.” Gilson, S.E.; Li, P.; Szymanowski, J.E.S.; White, J.; Ray, D.; Gagliardi, L.; Farha, O.K.; Burns, P.C.; Am. Chem. Soc.; 2019, 141, pp. 11842-11846. doi.org/10.1021/jacs.9b06187
- “Stabilization of an Unprecedented Hexanuclear Secondary Building Unit in a Thorium-Based Metal-Organic Framework.” Li, P.; Goswami, S.; Otake, K.; Wang, X.; Chen, Z.; Hanna, S.L.; Farha, O.K.; Chem.; 2019, 58, pp. 3586-3590. doi.org/10.1021/acs.inorgchem.8b03511
- “Bottom-up construction of a superstructure in a porous uranium-organic crystal” Li, P.; Vermeulen, N. A.; Malliakas, C. D.; Gómez-Gualdrón, D. A.; Howarth, A. J.; Mehdi, B. L.; Dohnalkova, A.; Browning, N. D.; O’Keeffe, M.; Farha, O. K., Science 2017, 356, 624-627. doi.org/10.1126/science.aam7851
- “Design and Synthesis of a Water-Stable Anionic Uranium-Based Metal–Organic Framework (MOF) with Ultra Large Pores” Li, P.; Vermeulen, N. A.; Gong, X.; Malliakas, C. D.; Stoddart, J. F.; Hupp, J. T.; Farha, O. K., Angew. Chem. 2016,128, 10514-10518. doi.org/10.1002/ange.201605547
Heterogeneous Catalysis for Energy-Related Applications
Catalysts composed of small metal/metal-oxide crystallites, well dispersed on porous solid materials, play essential roles in a variety of industrial gas-phase catalytic reactions including steam reforming, Fischer–Tropsch, and Haber–Bosch processes. As compared to conventional supports, the structures of MOFs, a class of porous materials comprised of organic linkers and inorganic nodes, are more readily tuned by rational ligand design. Furthermore, using crystalline MOFs as supports provides opportunities for atomically precise structural characterization of both active sites and supports, thereby facilitating detailed mechanistic studies of reactions.
In the Farha group, two deposition techniques are used to anchor catalytically active metal ions to MOFs:
1) solvothermal deposition in a MOF (SIM) and
2) atomic layer deposition in a MOF (AIM).
New MOF designs with other functionalities and enhanced stabilities are also being pursued.
The targeted catalysis is mainly gas-phase transformations, including alkene hydrogenation and oligomerization, alkane oxidative dehydrogenation, and selective oxidation of alkanes to alcohols. Collaborating with beamline scientists at Argonne National Lab and theorists, we correlate the catalytic activity/product distribution with active-site structures to lay out the design rules for the discovery of improved catalysts.
Selected Reviews
- “Using Nature’s Blueprint to Enable Catalysis with Earth-Abundant Metals,” R. M. Bullock, J. G. Chen, L. Gagliardi, P. J. Chirik, O. K. Farha, C. H. Hendon, C. W. Jones, J. A. Keith, J. Klosin, S. D. Minteer, R. H. Morris, A. T. Radosevich, T. B. Rauchfuss, N. A. Strotman, A. Vojvodic, T. R. Ward, J. Y. Yang and Y. Surendranath, Science, 2020, 369, Issue 6505, eabc3183. DOI: 10.1126/science.abc3183
- “Metal–Organic Framework Nodes as a Supporting Platform for Tailoring the Activity of Metal Catalysts.” Syed, Z. H.; Sha, F.; Zhang, X.; Kaphan, D. M.; Delferro, M.; Farha, O. K. ACS Catal. 2020, 10, 11556-11566. doi.org/10.1021/acscatal.0c03056
- “Metal-Organic Frameworks: A Tunable Platform to Access Single-Site Heterogeneous Catalysts.” Wasson, M.C.; Buru, C.T.; Chen, Z.; Islamoglu, T.; Farha, O.K.; Applied Catalysis A, General; 2019, 586, pp. 117214. doi.org/10.1016/j.apcata.2019.117214
- “Catalytic Zirconium/Hafnium-Based Metal–Organic Frameworks.” Rimoldi, M.; Howarth, A. J.; DeStefano, M. R.; Lin, L.; Goswami, S.; Li, P.; Hupp, J. T.; Farha, O. K. ACS Catal. 2017, 7, 997-1014. doi.org/10.1021/acscatal.6b02923
- “Postsynthetic Tuning of Metal–Organic Frameworks for Targeted Applications” Islamoglu, T.; Goswami, S.; Li, Z.; Howarth, A. J.; Farha, O. K.; Hupp, J. T.; Acc. Chem. Res.; 2017, 50, pp. 805-813 doi.org/10.1021/acs.accounts.6b00577
- “NanoMOFs: little crystallites for substantial applications” Majewski, M. B.; Noh, H.; Islamoglu, T.; Farha, O. K.; Mat. Chem. A; 2018, 6, pp. 7338-7350 doi.org/10.1039/C8TA02132E
Selected Research Articles
-
“Insights into the Structure–Activity Relationship in Aerobic Alcohol Oxidation over a Metal–Organic-Framework-Supported Molybdenum(VI) Catalyst” Chen, Y.; Zhang, X.; Wang, X.; Drout, R.J.; Mian, M.R.; Cao, R.; Ma, K.; Xia, Q.; Li, Z.; Farha, O.K. J. Am. Chem. Soc., 2021, 143, pp. 4302-4310 doi.org/10.1021/jacs.0c12963
-
“Insights into the Structure–Activity Relationships in Metal–Organic Framework-Supported Nickel Catalysts for Ethylene Hydrogenation” Wang, X.; Zhang, X.; Pandharkar, R.; Lyu, J.; Ray, D.; Yang, Y.; Kato, S.; Liu, J.; Wasson, M.C.; Islamoglu, T.; Li, Z.; Hupp, J.T.; Cramer, C.J.; Gagliardi, L.; Farha, O.K. ACS Catal., 2020, 10, pp. 8995-9005 doi.org/10.1021/acscatal.0c01844
-
“Vanadium Catalyst on Isostructural Transition Metal, Lanthanide, and Actinide Based Metal-Organic Frameworks for Alcohol Oxidation” Wang, X.; Zhang, X.; Li, P.; Otake, K.I.; Cui, Y.; Lyu, J.; Krzyaniak, M.D.; Zhang, Y.; Li, Z.; Liu, J.; Buru, C.T.; Islamoglu, T.; Wasielewski, M.R.; Li, Z.; Farha, O.K.; Am. Chem. Soc.; 2019, 141, pp. 8306-8314 doi.org/10.1021/jacs.9b02603
- “Catalytic Chemoselective Functionalization of Methane in a Metal-Organic Framework” Zhang, X.; Huang, Z.; Ferrandon, M.; Yang, D.; Robison, L.; Li, P.; C Wang, T. C.; Delferro, M.; Farha, O. K.; Nature Catalysis; 2018, 1, pp. 356-362 org/10.1038/s41929-018-0069-6
- “Single-Atom-Based Vanadium Oxide Catalysts Supported on Metal-Organic Frameworks: Selective Alcohol Oxidation and Structure Activity Relationship” Otake, K.; Cui, Y.; Buru, C. T.; Li, Z.; Hupp, J. T.; Farha, O. K.; Am. Chem. Soc.; 2018, 140, pp. 8652-8656 doi.org/10.1021/jacs.8b05107
- “Pushing the Limits on Metal-Organic Frameworks as a Catalyst Support: NU-1000 Supported Tungsten Catalysts for o-Xylene Isomerization and Disproportionation.” Ahn, S.; Nauert, S. L.; Buru, C. T.; Rimoldi, M.; Choi, H.; Schweitzer, N. M.; Hupp, J. T.; Farha, O. K.; Notestein, J. M.; Am. Chem. Soc.;2018, 140, pp. 8535-8543. doi.org/10.1021/jacs.8b04059
Water Purification
Clean water is becoming increasingly scarce and it is imperative we develop cost-efficient technologies capable of remediating large bodies of contaminated water. Rapid industrialization and agricultural growth in nations with minimal environmental regulations have caused extensive pollution. The presence of heavy metals such as lead and arsenic in what supplies is unacceptably common. And after several decades, the global environment is still recovering from the development, testing, and use of nuclear weapons during World War II and the Cold War.
In the Farha group, we are combating water contamination using metal-organic frameworks (MOFs) by capitalizing on unique functionality at 1) the metal node and 2) the organic linker. We also use MOFs as scaffolds to probe individual characteristics that contribute to selective contaminant capture. The permanent crystallinity of MOFs and the ability to grow high-quality crystals allows for the characterization of binding motifs and mechanisms by crystallographic techniques. We will continue to develop new materials with specific applications in water purification.
Selected Reviews
- “Zirconium Metal-Organic Frameworks for Organic Pollutant Adsorption.” Drout, R.J.; Robison, L.; Chen, Z.; Islamoglu, T.; Farha, O.K.; Trends in Chemistry 2019, 3, pp. 304-317. doi.org/10.1016/j.trechm.2019.03.010
Selected Research Articles
-
“Thermochemical Investigation of Oxyanion Coordination in a Zirconium-Based Metal–Organic Framework” Drout, R.J.; Gaidimas, M.A.; Farha, O.K. ACS Appl. Mater. Interfaces, 2021, Just Accepted Article doi.org/10.1021/acsami.1c05271
-
“Isothermal Titration Calorimetry to Explore the Parameter Space of Organophosphorus Agrochemical Adsorption in MOFs” Drout, R.J.; Kato, S.; Chen, H.; Son, F.A.; Otake, K.; Islamoglu, T.; Snurr, R.Q.; Farha, O.K. J. Am. Chem. Soc., 2020, 142, pp.12375-12366 doi.org/10.1021/jacs.0c04668
-
“Tuning the Atrazine Binding Sites in An Indium-Based Flexible Metal-Organic Framework” Chen, Y.; Zhang, X.; Chen, H.; Drout, R.J.; Chen, Z.; Mian, M.R.; Maldonado, R.R.; Ma, K.; Wang, X.; Xia, Q.; Li, Z.; Islamoglu, T.; Snurr, R.Q.; Farha, O.K. ACS Appl. Mater. Interfaces, 2020, 12, pp. 44762-44768 doi.org/10.1021/acsami.0c13022
- “Efficient Capture of Perrhenate and Pertechnetate by a Mesoporous Zr Metal–Organic Framework and Examination of Anion Binding Motifs” Drout, R. J.; Otake, K.; Howarth, A. J.; Islamoglu, T.; Zhu, L.; Xiao, C.; Wang, S.; Farha, O. K.; Mater.; 2018. 30, pp. 1277–1284 doi.org/10.1021/acs.chemmater.7b04619
- “Exploiting π-π Interactions to Design an Efficient Sorbent for Atrazine Removal from Water.” Akpinar, I.; Drout, R.J.; Islamoglu, T.; Kato, S.; Lyu, J.; Farha, O.K.; ACS Appl. Mater. Interfaces; 2019, 11, pp. 6097-6103. doi.org/10.1021/acsami.8b20355
- “Efficient extraction of inorganic selenium from water by a Zr metal-organic framework: investigation of volumetric uptake capacity and binding motifs.” Drout, R.J.; Howarth, A.J.; Otake, K.; Islamoglu, T.; Farha, O.K.; CrystEngComm.; 2018, 20, pp. 6140-6145. doi.org/10.1039/C8CE00992A
- “99-TcO (4-) remediation by a cationic polymeric network.” Li, J.; Dai, X.; Zhu, L.; Xu, C.; Zhang, D.; Silver, M. A.; Li, P.; Chen, L.; Li, Y.; Zuo, D.; Zhang, H.; Xiao, C.; Chen, J.; Diwu, J.; Farha, O. K.; Albrecht-Schmitt, T. E.; Chai, Z.; Wang, S.; Nature Communications; 2018, 9, 3007. doi.org/10.1038/s41467-018-05380-5
Gas Storage/Separation/Capture
Metal–organic frameworks (MOFs) are an intriguing class of hybrid materials that are built by assembling metal centers with organic linkers. Through judicious choice of components, materials with permanent nanoscale porosity can be prepared. Some of the most notable examples have large internal surface areas and ultralow densities, with uniform cavities and voids with pre-designed molecular dimensions. These intriguing properties make them excellent candidates for small molecule storage and separation applications. The precise knowledge about the molecular structure of MOFs makes it possible to use highly accurate computational modeling of the static and dynamic interactions of guest molecules in the MOF pores to calculate adsorption isotherms, binding energies, and transport behavior, in both predictive and explanative modes. In Farha group, we design and synthesize MOFs with desired properties to store gases such as methane, hydrogen, oxygen or carbon dioxide, or separate industrially relevant gases.
Selected Reviews
- “New directions in gas sorption and separation with MOFs: general discussion.” Addicoat, M.; Bennett, T.; Chapman, K.; Denysenko, D.; Dinca, M.; Doan, H.; Easun, T.; Eddaoudi, M.; Farha, O.; Gagliardi, L.; Haase, F.; Hajiahmadi Farmahini, A.; Hendon, C.; Jorge, M.; Kitagawa, S.; Lamberti, C.; Lee, J.-S. M.; Leus, K.; Li, J.; Lin, W.; Liu, X.; Lloyd, G.; Lu, C.; Ma, S.; Perez, J. P. H.; Ranocchiari, M.; Rosi, N.; Stassen, I.; Ting, V.; van der Veen, M.; Van Der Voort, P.; Vande Velde, C. M. L.; Volkmer, D.; Vornholt, S.; Walsh, A.; Yaghi, O. M.; Faraday Discuss.; 2017, 201, pp. 175-194
org/10.1039/C7FD90044A - “Metal-Organic Frameworks against Toxic Chemicals” Islamoglu, T.; Chen, Z.; Wasson, M.C.; Buru, C.T.; Kirlikovali, K.O.; Afrin, U.; Mian, M.R.; Farha, O.K.; Chem. Rev., 2020, 120, pp. 8130-8160 doi.org/10.1021/acs.chemrev.9b00828
Selected Research Articles
- “Tuning the Structural Flexibility for Multi-Responsive Gas Sorption in Isonicotinate-Based Metal−Organic Frameworks” Chen, Y.; Idrees, K.B.; Son, F.A.; Wang, X.; Chen, Z.; Xia, Q.; Li, Z.; Zhang, X.; Farha, O.K. ACS Appl. Mater. Interface, 2021, 13, pp. 16820-16827 doi.org/10.1021/acsami.1c00061
- “Balancing volumetric and gravimetric uptake in highly porous materials for clean energy” Chen, Z.; Li, P.; Anderson, R.; Wang, X.; Zhang, X.; Robison, L.; Redfern, L.R.; Moribe, S.; Islamoglu, T.; Gómez-Gualdrón, D.A.; Yildirim, T.; Stoddart, J.F.; Farha, O.K.; Science, 2020, 368, pp. 297-303 doi.org/10.1126/science.aaz8881
- “Tailoring Pore Aperture and Structural Defects in Zirconium-Based Metal-Organic Frameworks for Krypton/Xenon Separation
Idrees, K.B.; Chen, Z.; Zhang, X.; Mian, M.R.; Drout, R.J.; Islamoglu, T.; Farha, O.K.; Chem. Mater., 2020, 32, pp. 3776-3782 doi.org/10.1021/acs.chemmater.9b05048 “
-
Zirconium-Based Metal–Organic Framework with 9-Connected Nodes for Ammonia Capture.” Chen, Y.; Zhang, X.; Ma, K.; Chen, Z.; Wang, X.; Knapp, J.; Alayoglu, S.; Wang, F.; Xia, Q.; Li, Z.; Islamoglu, T.; Farha, O.K.; ACS Appl. Nano Mater.; 2019, 2, pp. 6098-6102. doi.org/10.1021/acsanm.9b01534
- “Modular Synthesis of Highly Porous Zr-MOFs Assembled from Simple Building Blocks for Oxygen Storage.” Lyu, J.; Zhang, X.; Chen, Z.; Anderson, R.; Wang, X.; Wasson, M.C.; Bai, P.; Guo, X.; Islamoglu, T.; Gómez-Gualdrón, D.A.; Farha, O.K.; ACS Appl. Mater. Interfaces; Just Accepted Article; 2019. doi.org/10.1021/acsami.9b14439
- “Ammonia Capture within Isoreticular Metal-Organic Frameworks with Rod Secondary Building Units.” Moribe, S.; Chen, Z.; Alayoglu, S.; Syed, Z.H.; Islamoglu, T.; Farha, O.K.; ACS Materials Lett.; 2019, 1, pp. 476-480. doi.org/10.1021/acsmaterialslett.9b00307
- “Benchmark Study of Hydrogen Storage in Metal–Organic Frameworks under Temperature and Pressure Swing Conditions” García-Holley, P.; Schweitzer, B.; Islamoglu, T.; Liu, Y.; Lin, L.; Rodriguez, S.; Weston, M. H.; Hupp, J. T.; Gómez-Gualdrón, D. A.; Yildirim, T.; Farha, O. K.; ACS Energy Letters; 2018, 3, pp. 748-754 doi.org/10.1021/acsenergylett.8b00154
- “Energy-based descriptors to rapidly predict hydrogen storage in metal-organic frameworks.” Bucior, B.J.; Bobbitt, N.S.; Islamoglu, T.; Goswami, S.; Gopalan, A.; Yildrim, T.; Farha, O.K.; Bagheri, N.; Snurr, R.Q.; Syst. Des. Eng.; 2019, 4, pp. 162-174.
doi.org/10.1039/C8ME00050F - “Computer-aided discovery of a metal–organic framework with superior oxygen uptake.” Moghadam, P. Z.; Islamoglu, T.; Goswami, S.; Exley, J.; Fantham, M.; Kaminski, C. F.; Snurr, R. Q.; Farha, O. K.; Fairen-Jimenez, D.; Nature Communications; 2018, 9, 1378. doi.org/10.1038/s41467-018-03892-8
Chemical Separation
Chemical separations typically leverage the different physical and chemical properties of the species in the mixture to isolate one compound. Some properties, like boiling point, have been used for centuries to purify compounds, but distillation is an energy-intensive and expensive technique on an industrial scale. MOFs have been shown to serve as capable agents for separation technology, often relying on changes in pore size or Lewis basic sites to isolate the desired molecules. We are interested in leveraging the tunability of MOFs to study how the three-dimensional structure of these materials impacts their performance in challenging, shape-selective separations. In addition, we are investigating “reactive-separations” that utilize MOF-based catalysts to simplify a ternary mixture to a binary mixture that can be separated based on traditional absorbance-based techniques.
Selected Research Articles
-
“Precise Control of Cu Nanoparticle Size and Catalytic Activity through Pore Templating in Zr Metal-Organic Frameworks” Mian, M.R.; Redfern, L.R.; Pratik, S.M.; Ray, D.; Liu, J.; Idrees, K.B.; Islamoglu, T.; Gagliardi, L.; Farha, O.K.; Chem. Mater., 2020, 32, pp. 3078-3086 doi.org/10.1021/acs.chemmater.0c00059
-
“Enhancing Four-Carbon Olefin Production from Acetylene over Copper Nanoparticles in Metal-Organic Frameworks” Redfern, L.R.; Lo, W.-S.; Dillingham, I.J.; Eatman, J.G.; Mian, M.R.; Tsung, C.-K.; Farha, O.K.; ACS Appl. Mater. Interfaces, 2020, 12, pp. 31496-31502 doi.org/10.1021/acsami.0c08244
-
“Isothermal Titration Calorimetry to Explore the Parameter Space of Organophosphorus Agrochemical Adsorption in MOFs” Drout, R.J.; Kato, S.; Chen, H.; Son, F.A.; Otake, K.; Islamoglu, T.; Snurr, R.Q.; Farha, O.K. J. Am. Chem. Soc., 2020, 142, pp.12375-12366 doi.org/10.1021/jacs.0c04668
- “Zirconium-Based Metal-Organic Frameworks for the Removal of Protein-Bound Uremic Toxin from Human Serum Albumin.” Kato, S.; Otake, K.; Chen, H.; Akpinar, I.; Buru, C.T.; Islamoglu, T.; Snurr, R.Q.; Farha, O.K.; J. Am. Chem. Soc.;2019, 141, pp. 2568-2576. doi.org/10.1021/jacs.8b12525
- “Highly Selective Acetylene Semihydrogenation Catalyzed by Cu Nanoparticles Supported in a Metal-Organic Framework” Redfern, L. R.; Li, Z.; Zhang, X.; Farha, O. K.; ACS Appl. Nano Mater.; 2018, 1, pp. 4413-4417 org/10.1021/acsanm.8b01397
- “Effect of ionic liquid on sugar-aromatic separation selectivity by metal-organic framework NU-1000 in aqueous solution.” Yabushita, M.; Papa, G.; Li, P.; Fukuoka, A.; Farha, O.K.; Simmons, B.A.; Katz, A.; Fuel Process. Technol.; Just Accepted Article, 2019. org/10.1016/j.fuproc.2019.106189
- “Tuning ethylene gas adsorption via metal node modulation: Cu-MOF-74 for a high ethylene deliverable capacity” Liao, Y.; Zhang, L.; Weston, M. H.; Morris, W.; Hupp, J. T.; Farha, O. K.; Chem. Commun.; 2017, 53, pp. 9376-9379 org/10.1039/C7CC04160H
Physical and Chemical Stability of MOFs
Despite the extensive pursuit of design rules that describe MOF structural and chemical stability, the physical stability of MOFs under high pressure (i.e. compression) and low pressure (i.e. vacuum) remains largely underexplored. Understanding how the multi-dimensional structure of these porous frameworks dictates their behavior under extreme pressures is both fundamentally important and practically relevant for applications requiring pellet formation. Recent efforts in the Farha lab set out to understand the role of the organic linker in the compression of MOFs at high pressures. We investigated the pressure-response of MOFs using a diamond anvil cell and in-situ PXRD at the Advanced Photon Source at Argonne National Lab. As we continue to explore the pressure-response of MOFs, we will continue to develop structure-property relationships centered around various MOF components. Understanding the impact of topology on structure stability will allow us to design better materials for a variety of applications, including CWA degradation.
Selected Reviews
- “Mechanical properties of metal-organic frameworks” Redfern, L.R.; Farha, O.K.; Chemical Science; 2019, 10, 10666. doi.org/10.1039/c9sc04249k
- “A historical overview of the activation and porosity of metal-organic frameworks” Zhang, X.; Chen, Z.; Liu, X.; Hanna, S.L.; Wang, X.; Taheri-Ledari, R.; Maleki, A.; Li, P.; Farha, O.K.; Chem. Soc. Rev., 2020, 49, 7406-7427 doi.org/10.1039/D0CS00997K
- “Chemical, thermal and mechanical stabilities of metal–organic frameworks” Howarth, A. J.; Liu, Y.; Li, P.; Li, Z.; Wang, T. C.; Hupp, J. T.; Farha, O. K.; Nature Reviews Materials; 2016, 1, pp. 15018 doi.org/10.1038/natrevmats.2015.18
Selected Research Articles
-
“Transient Catenation in a Zirconium-Based Metal–Organic Framework and Its Effect on Mechanical Stability and Sorption Properties” Robison, L.; Gong, X.; Evans, A.M.; Son, F.A.; Wang, X.; Redfern, L.R.; Wasson, M.C.; Syed, Z.H.; Chen, Z.; Idrees, K.B.; Islamoglu, T.; Delferro, M.; Dichtel, W.R.; Coudert, F.-X.; Gianneschi, N.C.; Farha, O.K. J. Am. Chem. Soc., 2021, 143, pp. 1503-1512 doi.org/10.1021/jacs.0c11266
-
“Isolating the Role of the Node-Linker Bond in the Compression of UiO-66 Metal-Organic Frameworks” Redfern, L.R.; Ducamp, M.; Wasson, M.C.; Robison, L.; Son, F.A.; Coudert, F.-X.; Farha, O.K.; Chem. Mater, 2020, 32, pp. 5864-5871 doi.org/10.1021/acs.chemmater.0c01922
-
“Designing Porous Materials to Resist Compression: Mechanical Reinforcement of a Zr-MOF with Structural Linkers” Robison, L.; Drout, R.J.; Redfern, L.R.; Son, F.A.; Wasson, M.C.; Goswami, S.; Chen, Z.; Olszewski, A.; Idrees, K.B.; Islamoglu, T.; Farha, O.K.; Chem. Mater., 2020, 32, pp.3545-3552. doi.org/10.1021/acs.chemmater.0c00634
-
“Porosity Dependence of Compression and Lattice Rigidity in Metal-Organic Framework Series” Redfern, L.R.; Robison, L.; Wasson, M.C.; Goswami, S.; Lyu, J.; Islamoglu, T.; Chapman, K.W.; Farha, O.K. Am. Chem. Soc.; 2019, 141, pp. 4365-4371 doi.org/10.1021/jacs.8b13009
- “Torsion Angle Effect on the Activation of UiO Metal-Organic Frameworks” Ayoub, G.; Islamoglu, T.; Goswami, S.; Friščić, T.; Farha, O.K.; ACS Appl. Mater. Interfaces; 2019, 11, pp. 15788-15794 org/10.1021/acsami.9b02764
- “Water stabilization of Zr6-based metal-organic frameworks via solvent-assisted ligand incorporation.” Deria, P.; Chung, Y. G.; Snurr, R. Q.; Hupp, J. T.; Farha, O. K., Chemical Science 2015, 6, 5172-5176. doi.org/10.1039/c5sc01784j
Characterization Tools for MOFs
In the Farha group, we are interested in establishing reliable and transferrable methods that allow us and the scientific community to advance our understanding of MOF formation process as well as interactions of the substrates with MOFs. Therefore, we have begun using isothermal titration calorimetry (ITC) to quantify the strength and type of interactions (e.g. coordination, H-bonding, hydrophobic effect) between analytes and sorbents. We utilized potentiometric titration on Zr-MOFs which allowed us to determine the proton topology, therefore, the defects present in these kinds of MOFs. In situ powder X-ray diffraction powder X-ray diffraction (PXRD) measurements accompanied by high resolution transmission electron microscopy (HR-TEM), we characterize not only the thermodynamic MOF products, but also the kinetic products formed during the synthesis.
Selected Research Articles
- “Thermochemical Investigation of Oxyanion Coordination in a Zirconium-Based Metal–Organic Framework” Drout, R.J.; Gaidimas, M.A.; Farha, O.K. ACS Appl. Mater. Interfaces, 2021, Just Accepted Article doi.org/10.1021/acsami.1c05271
- “Isothermal Titration Calorimetry to Explore the Parameter Space of Organophosphorus Agrochemical Adsorption in MOFs” Drout, R.J.; Kato, S.; Chen, H.; Son, F.A.; Otake, K.; Islamoglu, T.; Snurr, R.Q.; Farha, O.K. J. Am. Chem. Soc., 2020, 142, pp.12375-12366 doi.org/10.1021/jacs.0c04668
- “Isothermal Titration Calorimetry to Investigate Uremic Toxins Adsorbing onto Metal-Organic Frameworks.” Kato, S.; Drout, R. J.; Farha, O. K. Cell Reports Physical Science 2020, 1, 100006. doi.org/10.1016/j.xcrp.2019.100006
- “Interrogating Kinetic versus Thermodynamic Topologies of Metal-Organic Frameworks via Combined Transmission Electron Microscopy and X-ray Diffraction Analysis” Gong, X.; Noh, H.; Gianneschi, N.C.; Farha, O.K.; Am. Chem. Soc.; 2019, 141, pp. 6146-615. doi.org/10.1021/jacs.9b01789
- “Evaluation of Bronsted acidity and proton topology in Zr- and Hf-based metal-organic frameworks using potentiometric acid-base titration” Klet, R. C.; Liu, Y.; Wang, T. C.; Hupp, J. T.; Farha, O. K.; Mat. Chem. A 2016, 4, pp. 1479-1485 doi.org/10.1039/C5TA07687K
Porous Organic Polymers
In the Farha group, by means of new materials chemistry capabilities developed by our group, we design, engineer, and construct porous polymers for capturing toxic gases such as ammonia and sulfur dioxide as well as for water purification. Furthermore, we also design and synthesize highly reactive porous and processable polymers that can react with organo-phosphate/phosphonate nerve agents, sulfur mustard blistering agents, and their simulants for potential use in protective gear.
Selected Research Articles
- “An Amidoxime-Functionalized Porous Reactive Fiber against Toxic Chemicals” Jung, D.; Kirlikovali, K.O.; Chen, Z.; Idrees, K.B.; Atilgan, A.; Cao, R.; Islamoglu, T.; Farha, O.K., ACS Mater. Lett., 2021, 3, pp. 320-326 doi.org/10.1021/acsmaterialslett.0c00598
- “Postsynthetically Modified Polymers of Intrinsic Microporosity (PIMs) for Capturing Toxic Gases” Jung, D.; Chen, Z.; Alayoglu, S.; Mian, M.R.; Goetjen, T.A.; Idrees, K.B.; Kirlikovali, K.O.; Islamoglu, T.; Farha, O.K. ACS Appl. Mater. Interfaces, 2021, 13, pp. 10409–10415 doi.org/10.1021/acsami.0c21741
- “Reactive Porous Polymers for Detoxification of a Chemical Warfare Agent Simulant.” Jung, D.; Das, P.; Atilgan, A.; Li, P.; Hupp, J. T.; Islamoglu, T.; Kalow, J. A.; Farha, O. K. Chem. Mater. 2020, 32, 9299-9306. doi.org/10.1021/acs.chemmater.0c03160
Porous Clusters
In the Farha Group, we are interested in developing atomically precise porous assemblies of metallic oxo clusters as a stepping stone to studying catalytic activity complicated in ill-defined bulk metal oxides. Thus far, we have induced porous packing in cerium-oxo cluster assemblies through the judicious choice of a capping agent. With a wide breadth of instrumentation within the IMSERC and REACT core faciliites, we can further interrogate structure-activity relationships relevant to oxidative and reductive catalysis. We envision that other families of porous periodic clusters can be targeted through such bottom-up design strategies.

Selected Research Articles
“Supramolecular Porous Assemblies of Atomically Precise Catalytically Active Cerium-Based Clusters.” Wasson, M. C.; Zhang, X.; Otake, K.-i.; Rosen, A. S.; Alayoglu, S.; Krzyaniak, M. D.; Chen, Z.; Redfern, L. R.; Robison, L.; Son, F. A.; Chen, Y.; Islamoglu, T.; Notestein, J. M.; Snurr, R. Q.; Wasielewski, M. R.; Farha, O. K. Chem. Mater. 2020, 32, 8522-8529. doi.org/10.1021/acs.chemmater.0c02740
Composite Materials
The utilization of MOFs as well-defined crystalline supports is another emerging area of MOFs since they offer some superior properties compared to non-crystalline supports. The supported composite materials show improved stability and performance, sometimes synergistically. In the Farha group, we study the immobilization of polyoxometalates (POMs), nanoparticles, C60, carborane complexes etc either to stabilize the guest or to increase the performance of parent MOFs for various applications. Also, we use the pores of the MOFs to template the growth of metal nanoparticles to control nanoparticle diameter and spatial arrangement in these composites.
MOFs are often obtained in fine powders which challenges their implementation for commercial applications. To this end, we prepare composites of crystalline MOFs with flexible textile fibers to produce composites that retain the desired properties of each component. The ability to further tailor these composites’ shapes for incorporation into industrial equipment increases their potential in applications such as adsorption devices and protective gears.
Selected Reviews
- “Fiber Composites of Metal-Organic Frameworks” Ma, K.; Idrees, K.B.; Son, F.A.; Maldonado, R.; Wasson, M.C.; Zhang, X.; Wang, X.; Shehayeb, E.; Merhi, A.; Kaafarani, B.R.; Islamoglu, T.; Xin, J.H.; Farha, O.K.; Chem. Mater., 2020, 32, pp. 7120-7140 doi.org/10.1021/acs.chemmater.0c02379
-
“Metal–organic frameworks: A tunable platform to access single-site heterogeneous catalysts” Wasson, M.C.; Buru, C.T.; Chen, Z.; Islamoglu, T.; Farha, O.K.; Appl. Catal. A Gen., 2019, 586, Article 117214 doi.org/10.1016/j.apcata.2019.117214
-
“Strategies for Incorporating Catalytically Active Polyoxometalates in Metal–Organic Frameworks for Organic Transformations. ” Buru, C. T.; Farha, O. K. ACS Appl. Mater. Interfaces 2020, 12, 5345-5360. doi.org/10.1021/acsami.9b19785
-
“NanoMOFs: little crystallites for substantial applications” Majewski, M. B.; Noh, H.; Islamoglu, T.; Farha, O. K.; J. Mater. Chem. A, 2018, 6, pp. 7338-7350 doi.org/10.1039/C8TA02132E
-
Selected Research Articles
- “Near-instantaneous catalytic hydrolysis of organophosphorus nerve agents with zirconium-based metal-organic-framework hydrogel composites” Ma, K.; Wasson, M.C.; Wang X.; Zhang X.; Idrees, K.B.; Chen, Z.; Wu, Y.; Lee, S.-J.; Cao, R.; Chen, Y.; Yang, L.; Son, F.A.; Islamoglu, T.; Peterson, G.W.; Mahle, J.J.; Farha, O.K. Chem Catalysis, 2021, 1, pp. 1-13 doi.org/10.1016/j.checat.2021.06.008
-
“Enhancing Four-Carbon Olefin Production from Acetylene over Copper Nanoparticles in Metal–Organic Frameworks” Redfern, L. R.; Lo, W.-S.; Dillingham, I. J.; Eatman, J. G.; Mian, M. R.; Tsung, C.-K.; Farha, O. K. ACS Appl. Mater. Interfaces 2020, 12, 31496-31502 doi.org/10.1021/acsami.0c08244
-
“Integration of Metal–Organic Frameworks on Protective Layers for Destruction of Nerve Agents under Relevant Conditions” Chen, Z.; Ma, K.; Mahle, J.J.; Wang, H.; Syed, Z.H.; Atilgan, A.; Chen, Y.; Xin, J.H.; Islamoglu, T.; Peterson, G.W.; Farha, O.K.; J. Am. Chem. Soc., 2019, 141, pp. 20016-20021 doi.org/10.1021/jacs.9b11172
- “Benign Integration of a Zn-Azolate Metal−Organic Framework onto Textile Fiber for Ammonia Capture” Ran, C.; Chen, Z.; Chen, Y.; Idrees, K.B.; Hanna, S.L.; Wang, X.; Goetjen, T.A.; Sun, Q.; Islamoglu, T.; Farha, O.K. ACS Appl. Mater. Interfaces, 2020, 12, pp. 47747-47753 doi.org/10.1021/acsami.0c14316
- “Scalable and Template-Free Aqueous Synthesis of Zirconium-Based Metal–Organic Framework Coating on Textile Fiber” Ma, K.; Islamoglu, T.; Chen, Z.; Li, P.; Wasson, M.C.; Chen, Y.; Wang, Y.; Peterson, G.W.; Xin, J.H.; Farha, O.K.; J. Am. Chem. Soc.; 2019, 141, pp. 15626-15633 doi.org/10.1021/jacs.9b07301
- “ Increased Electrical Conductivity in a Mesoporous Metal–Organic Framework Featuring Metallacarboranes Guests. ” Kung, C.-W.; Otake, K.; Buru, C. T.; Goswami, S.; Cui, Y.; Hupp, J. T.; Spokoyny, A. M.; Farha, O. K.J. Am. Chem. Soc. 2018, 140, 3871-3875. doi.org/10.1021/jacs.8b00605
- “Adsorption of a Catalytically Accessible Polyoxometalate in a Mesoporous Channel-type Metal–Organic Framework.” Buru, C. T.; Li, P.; Mehdi, B. L.; Dohnalkova, A.; Platero-Prats, A. E.; Browning, N. D.; Chapman, K. W.; Hupp, J. T.; Farha, O. K. Chem. Mater. 2017, 29, 5174-5181. doi.org/10.1021/acs.chemmater.7b00750