Mission of the Synaptopathies Lab
The synaptic substrates of neuropsychiatric disorders
“Our mission is to uncover the molecular and cellular mechanisms whereby neurons communicate with each other within brain circuits, how they malfunction in neuropsychiatric disorders, and to use this knowledge to develop new therapies for such disorders.” –Peter Penzes

Brain & Behavior Research Foundation Award 2024
“Peter Penzes, Ph.D., Professor, Northwestern University, will continue with research in which his team has detected synaptic ectodomains in cerebrospinal fluid, a discovery with potential breakthrough implications for biomarkers and novel therapeutics in schizophrenia.”

Article : Autism therapeutics using IGFBP2 peptides : Molecular Psychiatry 2023
“We identified multiple disease-relevant phenotypes in a Shank3 heterozygous mouse and showed that JB2 rescued deficits in synaptic function and plasticity, learning and memory, ultrasonic vocalizations, and motor function; it also normalized neuronal excitability and seizure susceptibility. Notably, JB2 rescued deficits in the auditory evoked response latency, alpha peak frequency, and steady-state electroencephalography response, measures with direct translational value to human subjects. These data demonstrate that JB2 is a potent modulator of neuroplasticity with therapeutic potential for the treatment of PMS and ASD.”

Article : Genetic correction of 16p11.2 duplication syndrome, Nature communications 2023
Using gene co-expression and interactome analysis, we show that PRRT2 is a major hub in the epilepsy subnetwork. Remarkably, correcting Prrt2 copy number rescued aberrant circuit properties, seizure susceptibility and social deficits in 16p11.2dup/+ mice. We show that proteomics and network biology can identify important disease hubs in multigenic disorders, and reveal mechanisms relevant to the complex symptomatology of 16p11.2 duplication carriers.

Article : IPSC and 16p11-schizophrenia on Biological Psychiatry 2023
“Our results indicate the presence of 16p11.2 duplication-dependent alterations in SCZ patient–derived iENs. Transcriptomics and cellular phenotyping reveal overlap between isogenic and patient-derived iENs, suggesting a central role of glutamatergic, morphological, and calcium dysregulation in 16p11.2 duplication-mediated pathogenesis. Moreover, excitatory dysfunction during early neurodevelopment is implicated as the basis of SCZ pathogenesis in 16p11.2 duplication carriers. Our results support network synchrony and calcium handling as outcomes directly linked to this genetic risk variant.”

Article : A developmental delay linked missense mutation in Kalirin-7 disrupts protein function and neuronal morphology
Together these results indicate that reduced Rac1-GEF activity as result of E1577K mutation impairs neuroarchitecture, connectivity and NMDAr activity, and is a likely contributor
to impaired neurodevelopment in a patient with developmental delay.

Article : Lithium rescues dendritic abnormalities in Ank3, Neuropsychopharmacology 2023
Altogether, our results suggest that dendritic abnormalities observed in loss of
function ANK3 variants and BD patients may be rescued by lithium treatment. Additionally, drugs selectively targeting GSK3β and cAMP pathways could be beneficial in BD.

Review : Spinules, aging and psychiatric disorders, in Frontiers 2023
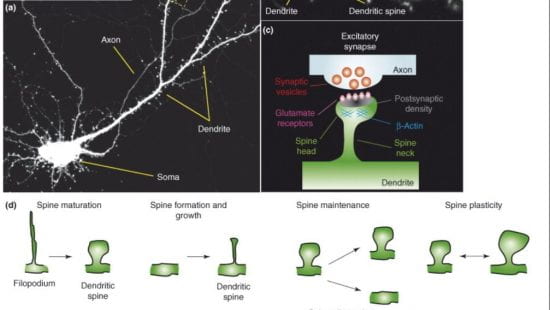
Dendritic spines are highly dynamic and changes in their density, size, and shape underlie structural synaptic plasticity in cognition and memory. Fine membranous protrusions of spines, termed dendritic spinules, can contact neighboring neurons or glial cells and are positively regulated by neuronal activity. Spinules are thinner than filopodia, variable in length, and often emerge from large mushroom spines.
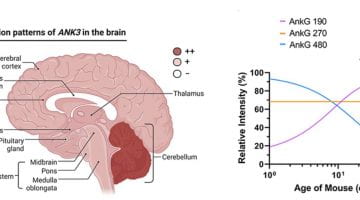
New Review 2022 : Roles and mechanisms of ankyrin-G in neuropsychiatric disorders
In this review, we describe the expression patterns of ankyrin-G isoforms, which vary according to the stage of brain development, and consider their functional differences. Furthermore, we discuss how posttranslational modifications of ankyrin-G affect its protein expression, interactions, and subcellular localization.
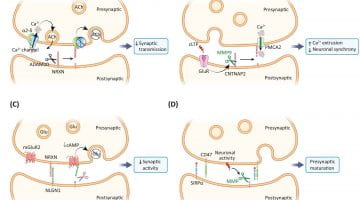
Trends In Neurosci. Review 2022 ! - Intercellular signaling by ectodomain shedding at the synapse
-Numerous synaptic proteins undergo ectodomain shedding (ES), often regulated by neuronal activity, and many more await discovery.
-Previously thought of as a modality for protein inactivation, emerging evidence indicates that ES generates soluble ectodomains which act as paracrine signals to modulate neuronal development, function, plasticity, and circuit properties.
-Faulty neuronal ES is associated with a growing number of neurological disorders.
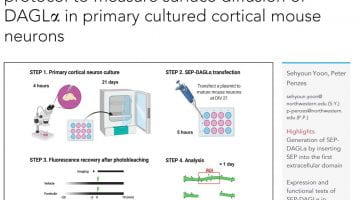
Star Protocol 2022 : A fluorescence recovery after photobleaching protocol to measure surface diffusion of DAGLα in primary cultured cortical mouse neurons
Highlights :
-Generation of SEP-DAGLα by inserting SEP into the first extracellular domain
-Expression and functional tests of SEP-DAGLα in HEK293T cells
-FRAP of SEP-DAGLα to determine DAGLα diffusion in dendritic shafts
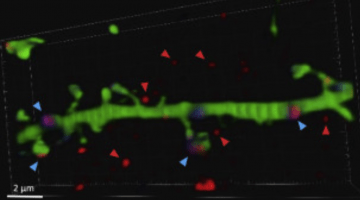
New Paper ! CNTNAP2 in CSF regulates calcium and Network activity in the brain, Neuron 2021 (Cell Press)
Highlights
• The neuronal sheddome is enriched in neurodevelopmental disorder risk factors
• CNTNAP2 is cleaved, and its ectodomain is detectable in the cerebrospinal fluid
• Neuron activity boosts CNTNAP2 shedding and the ectodomain binds to Ca2+ pump PMCA2
• CNTNAP2 ectodomain enhances Ca2+ extrusion and reduces neuronal network synchrony
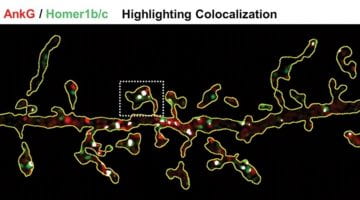
New Paper 2021 in Mol. Psychiatry : Homer1 promotes dendritic spine growth through ankyrin-G and its loss reshapes the synaptic proteome
Learn more about Homer1 regulation of synaptic plasticity through Ankyrin-G published in Molecular psychiatry !
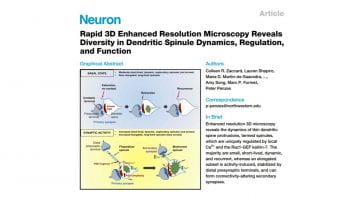
Rapid 3D Enhanced Resolution Microscopy Reveals Diversity in Dendritic Spinule Dynamics, Regulation, and Function
In brief :
Enhanced resolution 3D microscopy reveals the dynamics of thin dendritic spine protrusions, termed spinules,which are uniquely regulated by local Ca2+ and the Rac1-GEF kalirin-7. The majority are small, short-lived, dynamic, and recurrent, whereas an elongated subset is activity-induced, stabilized by distal presynaptic terminals, and can form connectivity-altering secondary synapses.
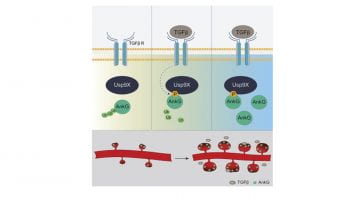
New paper published on Cell Reports from the Penzes lab !
Highlights :
Usp9X regulates ankyrin-G stability downstream of TGF-Beta to maintain spines
TGF-Beta-induced phosphorylation of Usp9X increases its interaction with ankyrin-G
In situ PLA/SIM reveal the spatial regulation of Usp9X/ankyrin-G interactions in spines
TGF-Beta is a potential therapeutic lead to correct synaptic deficits
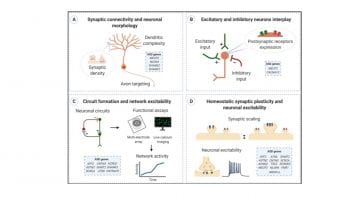
New Review : Excitation/Inhibition Imbalance in Human iPSC-derived Models of Autism spectrum disorder
Check out our new review published on Molecular Autism 2020 :
Here, we aim to review the latest advances in studying the different cellular and molecular mechanisms contributing to E/I balance using iPSC-based in vitro models of ASD.

Usp9X Controls Ankyrin-Repeat Domain Protein Homeostasis during Dendritic Spine Development
Check out our new exciting paper published in Neuron :
Yoon et al. show that deubiquitination of proteins containing ankyrin-repeat domains is essential for the correct developmental trajectory of cortical synapses, with disruption of the deubiquitinase Usp9X resulting in deficient synaptic structural plasticity as well as behavioral and clinical abnormalities.
Defining the Biology of Autism
“Peter Penzes, PhD, says the field of autism neurobiology is ripe for discovery and his team at the new Center for Autism and Neurodevelopment at Feinberg is laying the groundwork for new treatments for the disorder.”

New model of Alzheimer's disease
From the earliest changes in brain chemistry to novel approaches for clinical care, Northwestern scientists investigate a model spectrum of this devastating disease.

New Center for Autism and Neurodevelopment
The mission of the Center is to catalyze scientific collaborations aimed at understanding the biological bases of autism and related neurodevelopmental disorders, and to translate those findings into new treatments.
Diseases of interest.
Autism spectrum disorder, intellectual disability, epilepsy, schizophrenia, bipolar disorder, rare and orphan disorders, Alzheimer’s disease.
Synaptic substrates of neuropsychiatric disorders.
Recent exciting development in human genomics have uncovered myriads of rare mutations and common variants associated with increased risk for neuropsychiatric disorders. Globally, these gene sets are highly enriched in synaptic molecules, pointing to synapses are being one of the key cellular substrates in their pathologies. However, progress in translating such genetic richness into therapeutics is hampered by the lack of knowledge about the functions of many such disease risk factors. For this reason, we are investigating the synaptic functions and pathogenic mechanisms of a range of important risk factors for autism (ANK2, CNTNAP2, 16p11.2, SHANK3), intellectual disability (CASK), epilepsy (PRRT2), schizophrenia and bipolar disorder (ANK3, CACNA1C), and late-onset Alzheimer’s disease (BIN1).
Mechanisms of excitatory/inhibitory balance in the brain; comorbidity of neurodevelopmental disorders with epilepsy.
Epilepsy and seizure disorders often co-occur with autism and intellectual disability. For example, autism has a ~30% comorbidity with epilepsy. The mechanisms underlying this comorbidity is not well understood. Shared genetic risk factors, associated with both epilepsy and neurodevelopmental disorders, could provide an entry point into investigations of such mechanisms. In recent work we have uncovered mechanisms whereby neurodevelopmental disorder risk factors, such as CNTNAP2, ANK2, 16p11.2 also regulate ion channel homeostasis and excitatory/inhibitory balance in neuronal circuits.
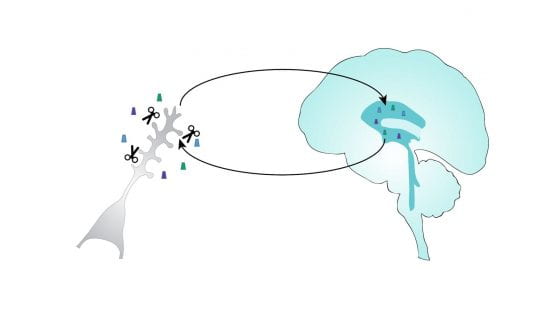
The synaptic sheddome.
Transmembrane proteins sometimes undergo ectodomain shedding upon proteolytic cleavage, but only a few such cases have been studied. Using unbiased global proteomic approaches, we have discovered that a large number of neuronal membrane proteins undergo ectodomain shedding. This sheddome is detectable in the cerebrospinal fluid and is enriched in synaptic proteins and disease risk factors. Unexpectedly, such ectodomains have novel biological functions in regulating cellular and network-level properties of brain circuits. These studies aim to understand the biological functions of the synaptic sheddome and use this knowledge to develop new therapeutics and biomarkers.
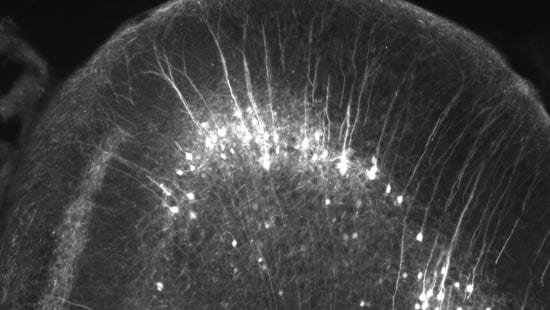
Cellular structural plasticity in the brain: dendrites, spines, spinules, tunneling nanotubes.
The nervous system is highly plastic and undergoes structural and functional changes during development, throughout the lifespan, and in neuropsychiatric disorders. Cellular and network architecture underlies normal brain wiring and rewiring, and is severely altered in diseases like autism, schizophrenia, bipolar disorder, and Alzheimer’s disease. Not only the neurons, but glial cells are also highly dynamic. Using advanced imaging approaches we are investigating the dynamics of brain cellular architecture during development, learning, stress, and disease.
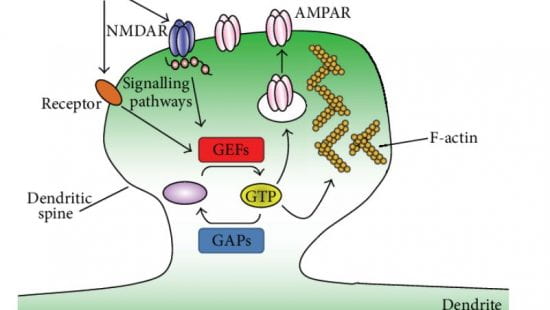
Drug development to target synaptic small GTPase pathways.
Small GTPases, such as Rac1 and Ras, have a wide range of biological functions throughout the body, including in the brain. In particular at synapses, GTPases and their direct upstream regulators, GEFs and GAPs, as well as their targets, are key regulators of development, maturation, plasticity. Not surprisingly, genes encoding proteins in this pathway are highly enriched in mutations in intellectual disability, autism, epilepsy, schizophrenia, and bipolar disease. In addition, expression levels of such proteins are reduced at synapses in schizophrenia and Alzheimer’s disease. Pharmacological targeting of these pathways could be desirable to reverse synaptic deficits in these and other disorders. However, this has proven to be very challenging in the past. Using novel high-throughput screening approaches, we are developing drug-like molecules to modulate the function of synaptic GEFs and GAPs, such as kalirin, Trio, and SynGAP1, to be further developed into therapeutics.
Methods
We employ a multi-disciplinary and multi-level combination of methodologies including super-resolution-, multi-photon-, calcium-, and time-lapse imaging, mouse genetics and behavioral analysis, human iPSC-derived neurons, bioinformatics, neuroproteomics, high-throughput and high content screens.